Earth's Fresh Water Problem
Water is life. Not only is it absolutely essential for all living organisms in order to live and thrive, it is the very basis of civilization as we know it. Beyond the need for clean drinking water, fresh water is also indispensable for farmland irrigation. Little wonder then why, with the Earth's population continuing to grow, that many fear for our planet's fresh water supply.
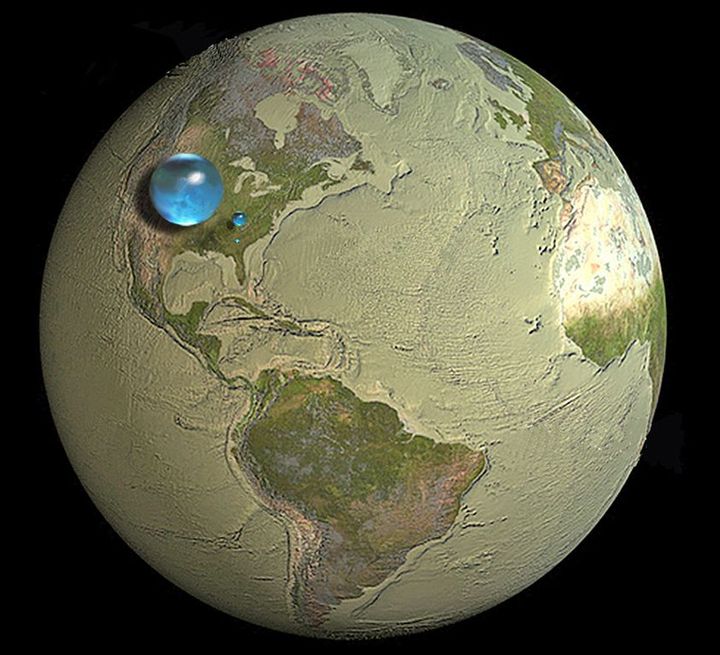
But if we were take the total amount of water contained on Earth and break it down according to what is drinkable and accessible, we would find just how limited this supply really is. Only 2.5% of the Earth's water is not salty, and the majority of that - 69% to be exact - takes the form of ice and glaciers contained within the Earth's Polar Regions.
In addition, another 1.8% of that exists below ground in the form of underground rivers and aquifers. This means that the amount of water that exists as groundwater, rivers, lakes, and streams - i.e. that which is immediately accessible for drinking and irrigation - is just 0.7%. Seen in this context, the limited and precious nature of freshwater becomes truly clear.
But what of the remaining 97.5% that takes the form of oceans and salt water? If it were possible to convert even a portion of this to fresh water, it would be possible to meet global demand indefinitely. As a result, researchers have been seeking cost-effective ways of turning sea water into drinking water for decades.
Current Desalination Efforts
According to the International Desalination Association (IDA), in June 2011, 15,988 desalination plants operated worldwide, producing 66.5 million cubic meters per day, providing water for 300 million people. However, the vast majority of this production took place within countries where access to freshwater is limited and cheaper alternatives (such as drilling for aquifers) are not available.
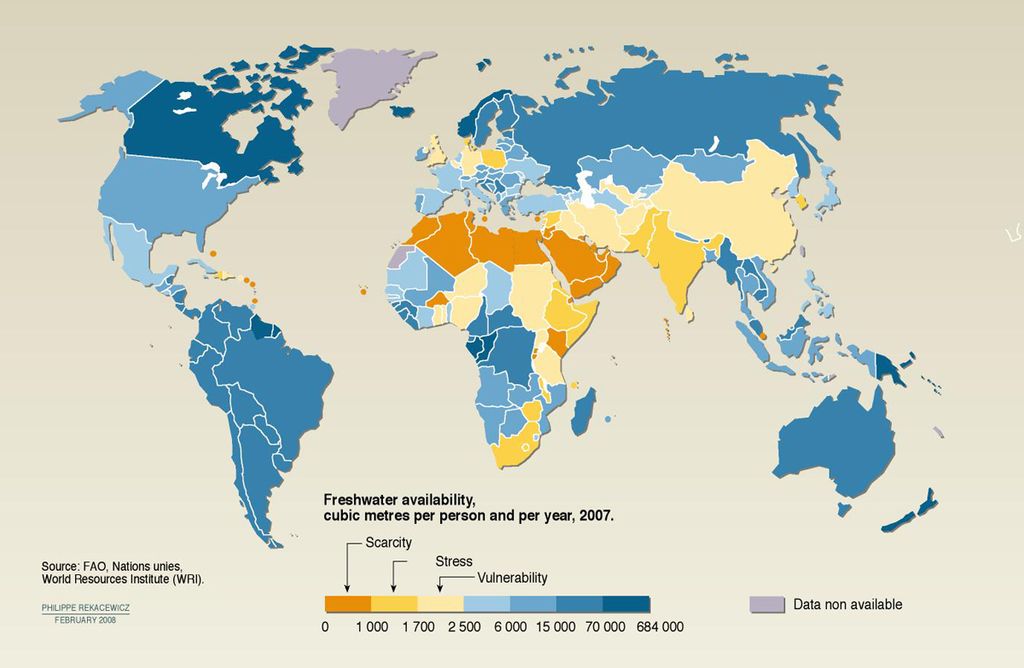
In addition, the National Intelligence Council, which reflects the combined input of 16 U.S. intelligence agencies, claims that global water demand will exceed sustainable supplies by 40 per cent by 2030. By this time, the world's population is expected to reach between 8 and 9 billion people, and it is predicted that Climate Change will have lead to the disappearance of water tables through diminished rainfall, changing weather patterns, and the drying up of riverbeds and lakes.
In short, demand will rise sharply as supply continues to drop. And given that current operations require a great deal of electricity and infrastructure to remain in operation, cheaper, more environmentally-sustainable measures need to be produced. Traditionally, the method of choice for desalination has been thermal distillation, where salt water is evaporated and the water is then captured and condensed to produce drinking water. This process has existed for centuries, and refinements in technology made it the only true means until the mid-20th century.

However, by the post-war era, scientists began to experiment with the osmotic process - aka. "reverse osmosis - which began to be adopted extensively. The first reported use of the term appeared in the US Department of Interior’s Office of Saline Water Commission 1955 annual report. Development continued, and in the 1970s, commercial membrane processes - such as reverse osmosis (RO) and electrodialysis (ED) - began to be used more extensively.
This process typically uses less energy than thermal distillation, which led to a reduction in overall desalination costs. This in turn led to the establishment of RO and ED operations in regions and municipalities all around the world where fresh water supplies were limited. By the 1980s and 90's, desalination technology became a fully commercial enterprise. At present, reverse osmosis (RO) accounts for approximately 60 percent of installed capacity.
In spite of this, desalination remains an energy intensive process, and future costs will continue to depend on the price of both energy and desalination technology. Hence why there is a push to come up with more cost-effective processes and to utilize renewable energy to power desalination plants. Environmental considerations are also a going concern, with operation's looking to reduce their carbon footprint.

Technology and New Desalination Methods
As a result, new methods are being considered which take advantage of new and emerging technologies. Electrodialysis Reversal (EDR) - where electricity is applied to salt water to neutralize chloride ions - is one such process. A fitting example of this is the seawater-desalinating chip: a small, microchannel device developed by researchers at the University of Texas at Austin and Germany’s University of Marburg.
Utilizing a microchannel that branches in two and an electrode embedded at the branching point, the chip applies a 3-volt charge to neutralize chloride ions, diverting the salt to one branch while allowing the water to continue down the other. The chip was developed in conjunction with Okeanos Technologies, who are now developing it as alternative to thermal distillation, reverse osmosis and more costly-versions of EDR.
Forward osmosis is another possibility, a process which GreenCentre Canada - a government-funded green chemistry research lab based at Queen’s University in Kingston, Ontario - is heavily invested in. Compared to reverse-osmosis, which requires a great deal of pressure to remove salt from water, forward osmosis uses a super-concentrated brine solution known as "switchable salts" to pull pure water from sea water and deposit it on the other side of a membrane.

The super-briny additive is then removed by forcing air that is heating to 50 °C through the solution, a process which alters the chemical properties of the salt additive and turns it into a gaseous form that rises out of the solution. This gas can then be collected and reused to create the next batch of super-concentrated brine solution, creating a sustainable loop that depends on relatively little input of outside energy.
And then there's low temperature distillation, a process exemplified by Vancouver-based Saltworks Technologies. In this process, waste heat or solar heat is used to assist specially tuned filters that selectively block the natural flow of sodium, chlorine and other ions as they move through various stages of concentration. The approach requires little pressure, making it tremendously energy-efficient when compared to conventional methods of salt removal such as distillation and reverse-osmosis.
Other innovative and experimental processes that are currently under consideration include membrane distillation, pressure-retarded osmosis, biomimetic and graphene membranes. Nations that are at the forefront of implementation include Australia, Israel, Saudi Arabia, and the south-western United States, where current or impending demand for fresh water (combined with scarcity) drives demand and is spurring development.
Looking to the future, the IDA’s Energy Task Force has specifically challenged the desalination industry to achieve a further reduction of 20% in energy requirements for seawater desalination by 2015.
Sources:
- www.gizmag.com/desalination-water-chip/28172/
- water.usgs.gov/edu/drinkseawater.html
- idadesal.org/desalination-101/desalination-overview/
- www.okeanostech.com/technology/
- www.unep.org/dewa/vitalwater/
- publicintelligence.net/global-trends-2030/
- science.howstuffworks.com/environmental/earth/oceanography/desalination.htm
- www.utexas.edu/news/2013/06/27/chemists-work-to-desalt-the-ocean-for-drinking-water-one-nanoliter-at-a-time/
- www.thestar.com/business/2013/01/18/new_approach_to_water_desalination_holds_great_potential_hamilton.html
Image Credits:
- Global Water Usage: USGS
- Global Water Stress: unep.org
- Limassol Desalination Plant, Cyprus: logicom.net
- Reverse Osmosis: Pearson Prentice Hall/roplant.org
- Desalination Chip: utexas.edu